 
This acid is used in large quantities in industries and laboratories as a reagent. Sulphuric acid is a strong acid that is formed by oxidizing solutions of sulphur dioxide. But before proceeding to the questions and their relevant answer, first, let us introduce you to sulphuric acid and copper oxide. In this article, we will discuss and answer all the questions related to the reaction of copper oxide and sulphuric acid in detail. Given this information, determine the activation energy for the reverse reaction, Er, and comment on the significance of the value (one sentence only). (iii)The activation energy for the forward reaction, Ef, of step 1 (Equation 3) is 243.4 kJ mol^-1. (ii) With reference to the three-step mechanism (Equations 3–5 ), and assuming that the second step (Equation 4) is rate-limiting, derive the chemical rate equation for this mechanism and then compare it with the experimental rate equation given in Equation 2. (i) Explain why the form of the experimental rate equation indicates that Reaction 1 cannot be an elementary reaction. Under certain experimental conditions, the experimental rate equation was found to be:Ī three-step mechanism has been proposed for Reaction 1: 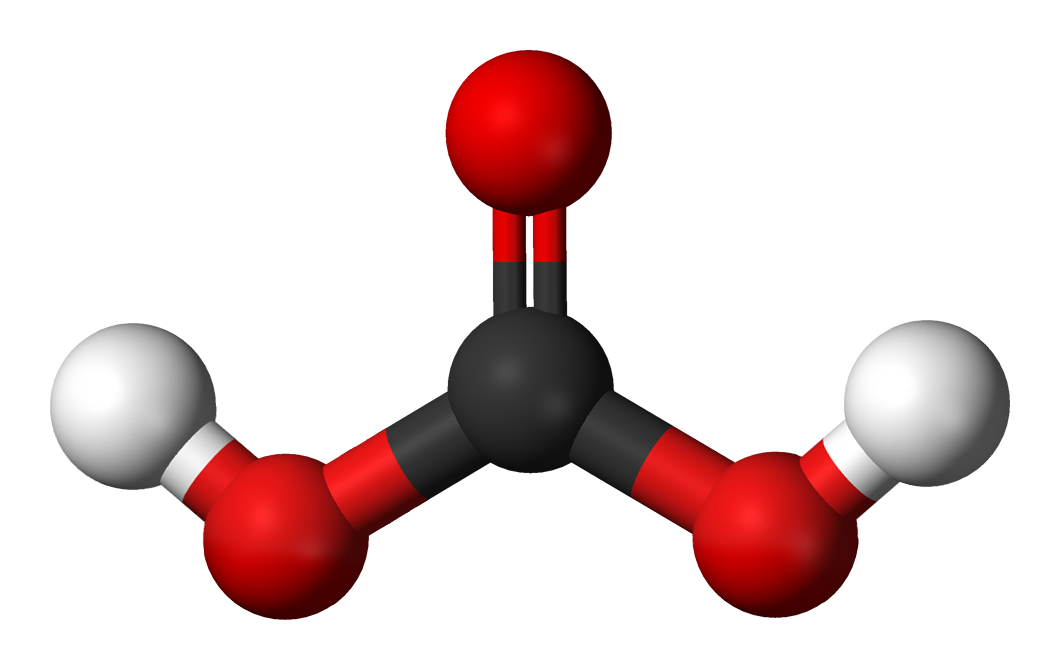
The gas-phase reaction between trichloromethane, CHCl3, and chlorine, Cl2, has time-independent stoichiometry and can be represented as follows:Įquation 1 CHCl3(g) + Cl2(g) = CCl4(g) + HCl(g) This chemistry is important in understanding how hard water is formed and then limescale is formed in kettles and hot water boilers. This happens as the carbon dioxide forms acidic carbonic acid when it dissolves in the water, the carbonic acid (H2CO3) reacts further with the calcium carbonate: CO2 + H2O -> H2CO3H2CO3 +CaCO3 -–> Ca(HCO3)2 Ca(HCO3)2 = calcium hydrogen carbonate which is soluble in water. Bubbling carbon dioxide through the solution for an extended period of time makes the solution become clear and colourless. This is because chalk is precipitating in the limewater. The characteristic test for CO2, is checking that the limewater is milky. The white milky suspension/precipitate is caused by the formation of calcium carbonate. The appearance of this solid makes the liquid appear ‘milky’. Calcium carbonate is chalk, and when it is produced, it precipitates and solid particles of chalk appear. The carbon dioxide and limewater react to produce calcium carbonate and water. Due to this fact, you will often see that limewater is used to detect the presence of carbon dioxide.Ĭarbon dioxide reacts with limewater to form calcium carbonate, which precipitates out of the solution. The white precipitate can be easily detected by the person conducting the experiment. Limewater is a calcium hydroxide solution that produces a white precipitate of calcium carbonate when it reacts with carbon dioxide. Limewater is used in experiments because it is the easiest way to detect the presence of $CO_2$ gas. The equation of this reaction is given below: When lime water and carbon dioxide reacts, calcium carbonate is generated along with the water. $Ca(OH)_$Īll of these reactions are reversible. What happens when you mix carbon dioxide and lime water? Written as an Equation This happens as the carbon dioxide forms acidic carbonic acid when it dissolves in the water, the carbonic acid (H2CO3) reacts further with the calcium carbonate. Bubbling carbon dioxide through the solution for an extended period of time makes the solution become clear and colorless. The characteristic carbon dioxide test, is checking that the limewater is milky. The carbon dioxide and limewater react to produce water in addition to the calcium carbonate. What happens when Lime Water reacts to Carbon Dioxide?Ĭarbon dioxide reacts with limewater to form calcium carbonate, which precipitates out of the solution. In this article, we have answered all the questions related to the reaction of lime water and $CO_2$. But one of its most noteworthy property is that it is used to absorb carbon dioxide from the air. Limewater is an aqueous solution of slaked lime and you will find it in antacids, medicines and lotions. 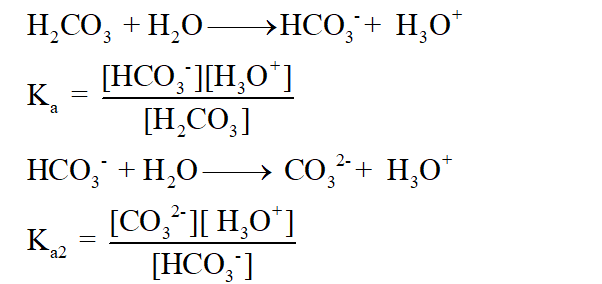
You may be wondering what is lime water used for. Carbon dioxide is the only gas that turns lime water cloudy. You may often come across a question "What gas turns limewater cloudy?" The answer to this question is well known. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
